Biologics Safety Testing Market Size to Hit US$ 8.8 Bn by 2033 | Persistence Market Research
Biologics safety testing ensures purity, quality, and compliance of advanced therapies, driven by innovation, strict regulations, and growing biologics demand.
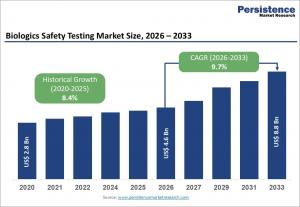
LONDON, UNITED KINGDOM, March 25, 2026 /EINPresswire.com/ -- The global biologics safety testing market is projected to reach US$ 4.6 billion in 2026 and expand to US$ 8.8 billion by 2033, growing at a CAGR of 9.7% during the forecast period. This market plays a vital role in ensuring the safety, purity, and effectiveness of biologics, which are increasingly used in the treatment of cancer, autoimmune diseases, and rare disorders. As biologics are derived from living cells using advanced technologies such as recombinant DNA, they require extensive testing to detect contaminants including viruses, endotoxins, mycoplasma, and residual host cell DNA. Stringent global regulatory frameworks and the growing complexity of biologics manufacturing are driving demand for reliable, standardized, and high-throughput safety testing solutions.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/18592
Market Dynamics
Driver: Surge in Biologics Approvals
The increasing number of biologics approvals, including vaccines, monoclonal antibodies, and cell and gene therapies, is a key factor driving the biologics safety testing market. Regulatory authorities require comprehensive testing throughout development and manufacturing stages, including sterility, viral clearance, and endotoxin testing. Each batch must meet strict quality standards before approval, increasing demand for validated assays and reagents. Accelerated approval pathways have further intensified the need for efficient and rapid testing methods that ensure safety without delaying product timelines. This surge in biologics development continues to strengthen the demand for advanced safety testing solutions.
Stringent Regulatory Mandates
Regulatory requirements remain a major force shaping the market. Authorities worldwide enforce strict guidelines for identity, potency, purity, and contamination control. Compliance with international standards is mandatory across all stages of biologics production. Testing for sterility, viral contamination, and residual impurities is essential, particularly for injectable and cell-based therapies. New methods such as recombinant Factor C assays are gaining popularity due to their improved sensitivity and reduced reliance on animal-derived materials. These evolving regulations are driving continuous investment in advanced technologies and validation processes.
Restraint: High Capital and Operational Costs
Despite strong growth prospects, high setup and operational costs present a significant challenge. Establishing compliant laboratories requires substantial investment in infrastructure, biosafety systems, and specialized equipment. Skilled personnel, ongoing validation, and regulatory compliance further increase operational expenses. Smaller companies and laboratories often face barriers in adopting advanced testing technologies due to financial constraints. Additionally, the transition to alternative testing methods requires additional validation and regulatory approval, adding to overall costs and complexity.
Opportunity: Growth in Cell and Gene Therapies
The rapid expansion of cell and gene therapy pipelines offers significant growth opportunities for the biologics safety testing market. These therapies require highly specialized testing methods to detect replication-competent viruses, residual DNA, and other contaminants. Due to their short shelf life, rapid testing solutions are essential to ensure timely product release. Automation and platform-based testing methods are gaining traction, improving efficiency and scalability. Increasing investments in advanced therapies and biomanufacturing infrastructure are expected to drive long-term demand for safety testing services.
Adoption of Rapid Microbiological Methods
The shift toward rapid microbiological methods is transforming the biologics safety testing landscape. Technologies such as PCR-based assays, automated systems, and advanced imaging techniques significantly reduce testing time while maintaining accuracy and compliance. These methods enable faster batch release and improved operational efficiency. Growing emphasis on reducing animal testing is also encouraging the adoption of alternative assays. Automation and robotics are increasingly used to handle large sample volumes, enhancing productivity and minimizing human error.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/18592
Category-wise Insights
Product Analysis
Kits and reagents dominate the biologics safety testing market due to their recurring usage and compatibility with high-throughput systems. These products are essential for routine testing procedures such as sterility, endotoxin, and mycoplasma detection. Their scalability and standardization make them widely adopted across both in-house and outsourced laboratories. Continuous innovation, including rapid and animal-free testing kits, is further supporting their growth.
Application Analysis
Vaccine and therapeutic development represent the largest application segment. Extensive testing is required throughout development and manufacturing to ensure safety and regulatory compliance. The growing demand for vaccines, biosimilars, and biologics has significantly increased testing volumes. As pharmaceutical companies focus on accelerating development timelines, the need for reliable safety testing continues to rise.
Regional Insights
North America holds the largest market share due to its advanced biopharmaceutical infrastructure, strong regulatory environment, and high investment in research and development. The presence of leading industry players and widespread adoption of advanced testing technologies further support market growth.
Asia Pacific is the fastest-growing region, driven by expanding biologics manufacturing, increasing healthcare investments, and supportive government initiatives. Countries such as China, India, and Japan are witnessing rapid growth in biopharmaceutical production and testing capabilities. The rise of contract research organizations and outsourcing activities is also contributing to market expansion.
Competitive Landscape
The biologics safety testing market is highly competitive, with key players focusing on technological advancements, strategic collaborations, and geographic expansion. Companies are investing in innovative testing solutions and expanding their service portfolios to meet evolving regulatory and industry demands. Continuous development of rapid, reliable, and cost-effective testing methods is expected to define the competitive landscape in the coming years.
Checkout Now & Download Complete Market Report: https://www.persistencemarketresearch.com/checkout/18592
Market Segmentation
Product
Kits & Reagents
Instruments
Services
Test Type
Endotoxin
Sterility
Cell Line Authentication and Characterization
Adventitious Agent Detection
Bioburden
Other
Application
Vaccine and Therapeutics Development
Blood and Blood-related Products Testing
Cellular and Gene Therapy
Tissue-related Products Testing
Stem Cell Research
Regions
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Read Related Reports:
Autoimmune Skin Diseases Treatment Market: The autoimmune skin diseases treatment market to grow from US$ 11.5 Bn in 2026 to US$ 29.6 Bn by 2033, expanding at a strong 14.5% CAGR worldwide.
Adult Hemoglobinopathy Testing Market: Global adult hemoglobinopathy testing market to grow from US$2.1 Bn in 2026 to US$3.4 Bn by 2033, registering a 5.5% CAGR driven by diagnostic advancements.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.